The Biogelis single-phase cross-linked hyaluronic acid range® already contains lidocaine in its formula.
Available in different concentrations and unique rheological characteristics, to meet the main needs of your patients, promoting everything from facial refinement to structuring with natural, long-lasting results.
Intradermal filling solution. For people who are feeling the first signs of time. Smoothes out marks, restoring a more youthful and natural appearance. Suitable for softening lines.
Anvisa registrations: Biogelis® Fine Lines with Lidocaine: 81872460011
Intradermal filling solution. Indicated for restoring volume to the mid-face, resulting in a smoother appearance and minimizing the impact of time.
Biogelis® Global with Lidocaine: 81872460009
Intradermal filling solution. It is a denser formula, suitable for adding volume to the chin and jaw, allowing lost volume to be recovered and restored. It improves the appearance, redefining the shape, contour and definition of the face.
Biogelis® Volume with Lidocaine: 81872460010
Intradermal filling solution. It is a denser formula, suitable for adding volume to the chin and jaw, allowing lost volume to be recovered and restored. It improves the appearance, redefining the shape, contour and definition of the face.
Biogelis® Volumax with Lidocaine: Anvisa Registration: 81872460013








Dynamic Natural Effect.
BIOGELIS® is of non-animal origin and was developed using our patented DNE technology® (Dynamic Natural Effect) which allows for a more homogeneous, cohesive gel with greater stability and durability³.
BIOGELIS® is single-phase with patented DNE technology® associated with a molecular structure with few free ends to bind to water. As a result, there is less edema post-procedure and the final volumization is practically the same as that observed at the time of application.
Immediately after application of Biogelis®If you are looking for a new look, you will see an instant improvement in the volume and contour of the treated area. The skin becomes firmer and more hydrated, while wrinkles and furrows are gently smoothed out, restoring natural volume and harmony to the face.
Post-procedure oedema is discreet and greatly reduced due to the high-tech manufacturing of the gel, which allows the final volumizing effect of the procedure to be practically the same as that observed at the time of application, bringing faithful and long-lasting results.

Check out the study in the Journal of Cosmetic Dermatology® on the use of hyaluronic acid to open up the eyes.
Dr. Elisete Crocco has developed an eye-opening filling technique using Pharmaesthetics Volume hyaluronic acid in the periorbital region. Recently published in the Journal of Cosmetic Dermtology.
"Fillers around the eyes are very difficult and risky. Anatomical knowledge, knowledge of the types of products that can be used, as well as their quality and quantity, and above all: common sense are essential."
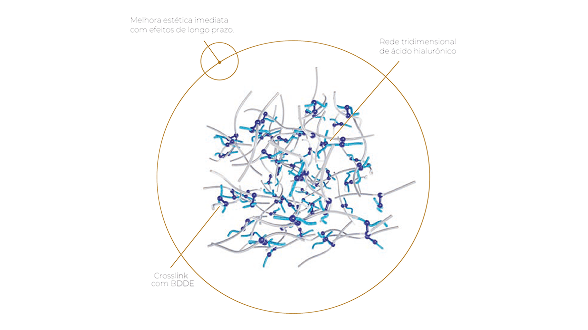
Use of the cross-linking agent BDDE in corrective medicine.
BDDE is the cross-linking agent most used today by the largest and best cross-linked hyaluronic acid producing laboratories in the world.
Understand why excellence in a treatment is directly related to the inflammatory response of a product in the tissues, and what the link is between this response and the cross-linking agent.
Environmental monitoring
Environmental monitoring in the Pharmaceutical Industry is the control of certain sequences of measurements and parameter indicators, aimed at preserving the best sanitary conditions in the manufacturing environment.
It is clear that in a sterile injectable drug industry the controls are stricter than in an industry of another risk class.
Rheology analysis of a hyaluronic acid gel
Learn more about the analytical process of rheology, the principles of how a rheometer works and how we obtain rheology results using the G' (better known as G Prime) and G" parameters.
Also understand the relationship between rheology and gel cohesiveness and how all these parameters are related.
Water for injections
Water purification, storage and distribution processes must ensure that pharmacopoeial specifications are met, maintained and controlled appropriately.
Water for injections is water that has undergone purification steps to remove all physicochemical and microbiological contaminants.
The degradation kinetics of hyaluronic acid
Knowing the importance of the degradation kinetics of a product based on cross-linked hyaluronic acid is fundamental and gives us the confidence to choose the best product.
An important concept measured by Degradation Kinetics is the durability of the hyaluronic acid gel in vivo and how this relates to cohesiveness, degree of cross-linking and resistance to degradation via hyaluronidase.
Visual inspection of hyaluronic acid
Visual inspection is a crucial stage in the manufacturing process of injectable products, in which it is possible to detect any easily identifiable visible defects in the containers (ampoules, syringes, etc.) and guarantee the constant quality of the product in terms of the absence of particulate matter, turbidity and, specifically in the case of fillers: air bubbles.
Full Face in 41-year-old patient
Follow a Full Face case carried out by dermatologist Giana Campoi, who used the different presentations of our single-phase hyaluronic acid gel on different parts of the face, according to the indication and result that the professional planned and wished to achieve - natural results without oedema.
Biogelis® is of non-animal origin and was developed using our patented DNE technology® (Dynamic Natural Effect) which allows for a more homogeneous, cohesive gel with greater stability and durability.